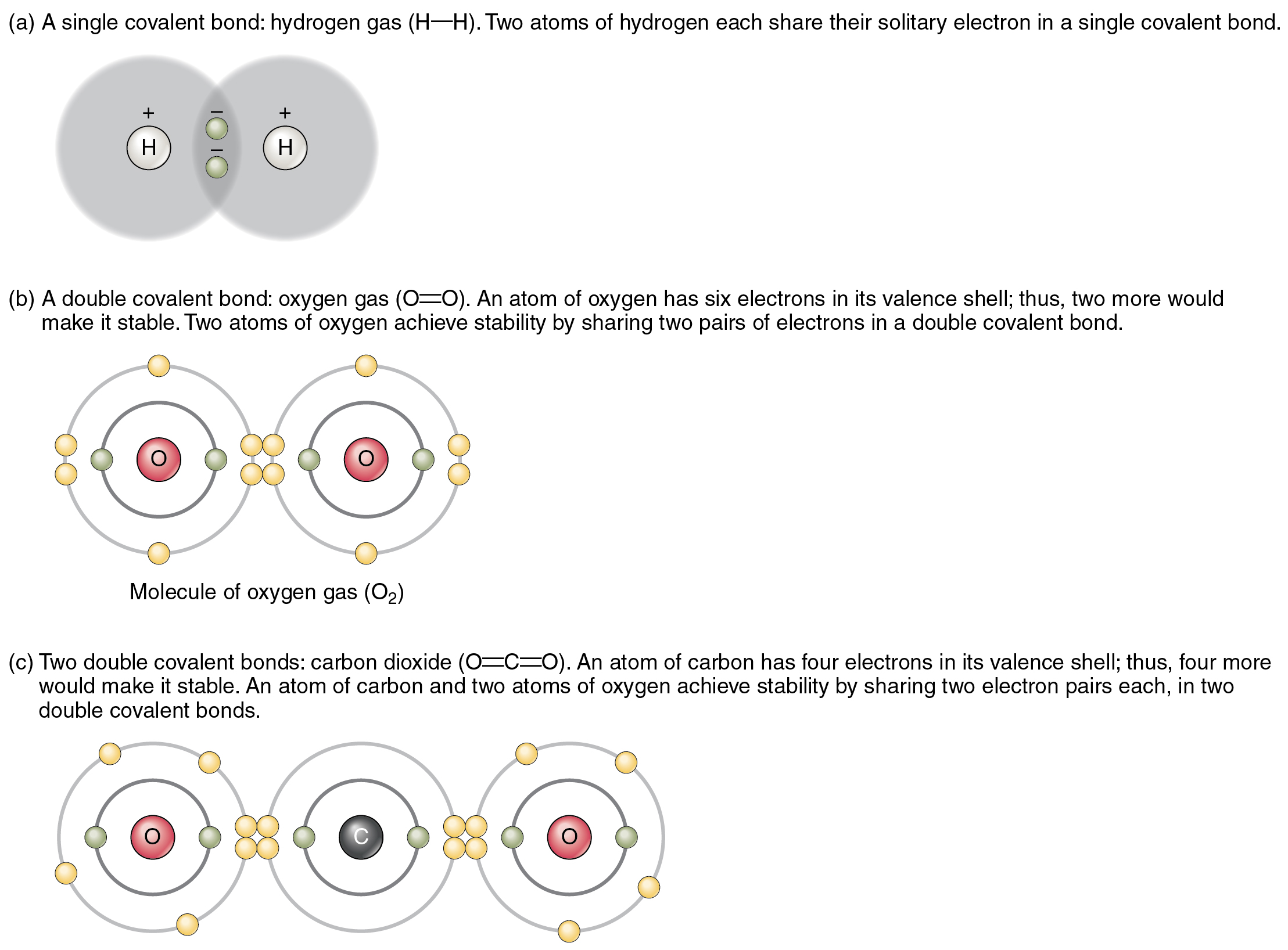
Which Element Is Most Likely To Form Three Covalent Bonds
Which Element Is Most Likely To Form Three Covalent Bonds - Covalent bonds form when two or more nonmetals combine. Thus, boron commonly forms three bonds, bh 3 , with a total of six electrons in the outermost shell. To obtain an octet, these atoms form three covalent bonds, as in nh 3 (ammonia). Web we refer to this as a pure covalent bond. Web the element which is most likely to form three covalent bonds is nitrogen. Which element is most likely to have a +i i i or −i i i oxidation state? Web if the atoms that form a covalent bond are identical, as in h 2, cl 2, and other diatomic molecules, then the electrons in the bond must be shared equally. The binding arises from the electrostatic attraction of their nuclei for the same electrons. B) si c) p d) s e) se 32). One lone pair and three unpaired electrons.
Web group 15 elements such as nitrogen have five valence electrons in the atomic lewis symbol: Web a chemical bond formed when two atoms share six electrons is a ________ bond; You'll get a detailed solution from a subject matter expert that helps you learn core concepts. The electrons involved are in the outer shells of the atoms. Web study with quizlet and memorize flashcards containing terms like which of the elements listed below would most likely form a polar covalent bond when bonded to oxygen?, which of the elements listed below would most likely form a nonpolar covalent bond when bonded to bromine?, define electronegativity: The binding arises from the electrostatic attraction of their nuclei for the same electrons. Web covalent bond, in chemistry, the interatomic linkage that results from the sharing of an electron pair between two atoms.
A rough estimate could also be done by simply looking at the periodic table. Web what elements make covalent bonds? Web which element is most. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Covalent bonding, in which neither atom loses complete control over its valence electrons, is much more common.
A) se b) si c) p d) s e) c Formation of an ionic bond by complete transfer of an electron from one atom to another is possible only for a fairly restricted set of elements. Web the electronegativity difference would be used to determine if an ionic or covalent bond is formed. Covalent bonding, in which neither atom loses complete control over its valence electrons, is much more common. Web what elements make covalent bonds? Web which element is most likely to form three covalent bonds?
Web study with quizlet and memorize flashcards containing terms like which of the elements listed below would most likely form a polar covalent bond when bonded to oxygen?, which of the elements listed below would most likely form a nonpolar covalent bond when bonded to bromine?, define electronegativity: Web a chemical bond formed when two atoms share six electrons is a ________ bond; In general, large differences in electronegativity result in ionic bonds, while smaller differences result in covalent bonds. Which element is most likely to have a +i i i or −i i i oxidation state? Electrons shared in pure covalent bonds have an equal probability of being near each nucleus.
Web what elements make covalent bonds? An atom that shares one or more of its electrons will complete its outer shell. Web the element carbon (c) is most likely to form covalent bonds with the element a. Thus, boron commonly forms three bonds, bh 3 , with a total of six electrons in the outermost shell.
Web Which Element Is Most Likely To Form Three Covalent Bonds?
A) se b) si c) p d) s e) c Web the electronegativity difference would be used to determine if an ionic or covalent bond is formed. Web covalent bonds usually form between nonmetals. Web a chemical bond formed when two atoms share six electrons is a ________ bond;
The Further Away Two Atoms Are From.
Web group 5a (15) elements such as nitrogen have five valence electrons in the atomic lewis symbol: Electrons shared in pure covalent bonds have an equal probability of being near each nucleus. Web which element is most. This problem has been solved!
There Are Compounds That Contain Both Covalent And Ionic Bonds, Such As Potassium Cyanide (Kcn) And Ammonium Chloride.
Web study with quizlet and memorize flashcards containing terms like which of the elements listed below would most likely form a polar covalent bond when bonded to oxygen?, which of the elements listed below would most likely form a nonpolar covalent bond when bonded to bromine?, define electronegativity: A) the number of each kind of atom must be the same on each side. P, c, s, si, se? Web what elements make covalent bonds?
Oxygen And Other Atoms In Group 16 Obtain An Octet By Forming Two Covalent Bonds:
Covalent bonding, in which neither atom loses complete control over its valence electrons, is much more common. To obtain an octet, these atoms form three covalent bonds, as in nh 3 (ammonia). To obtain an octet, these atoms form three covalent bonds, as in nh 3 (ammonia). Electrons shared in pure covalent bonds have an equal probability of being near each nucleus.