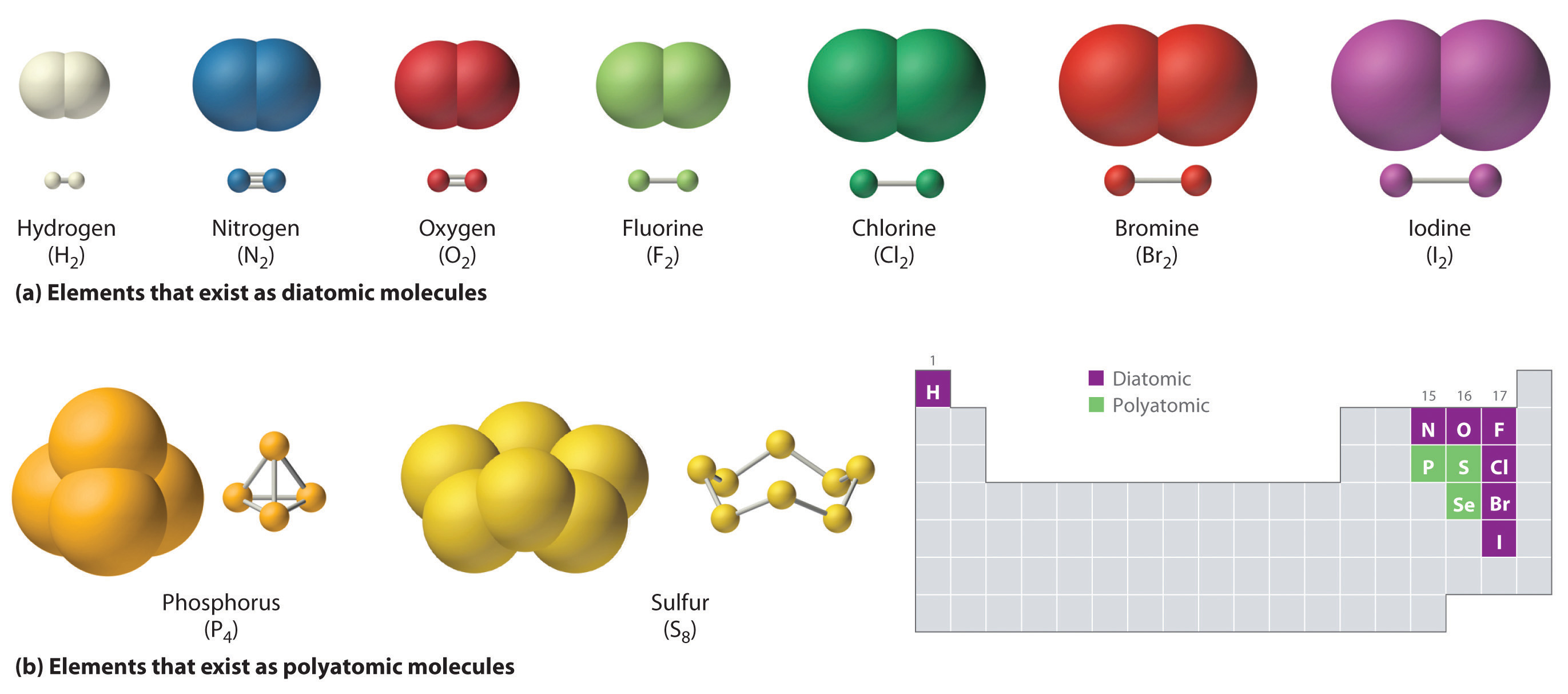
Which Combination Of Atoms Can Form A Polar Covalent Bond
Which Combination Of Atoms Can Form A Polar Covalent Bond - Na and h which of the following atoms has the highest first ionization energy? However, these polyatomic ions form ionic compounds by combining with ions of opposite charge. These atoms have the same electronegativity, resulting in a nonpolar covalent bond. Web which combination of atoms can form a polar covalent bond? Web which combination of atoms can form a polar covalent bond? Web compounds with polar covalent bonds have electrons that are shared unequally between the bonded atoms. Electronegativity difference should be low but shouldn't equal to zero. Some bonds between different elements are only minimally polar, while others are strongly polar. In a polar covalent bond, the electrons are unequally shared by the atoms and spend more time close to one atom than the other. Web a polar covalent bond is a bond formed when a shared pair of electrons are not shared equally.
H and br explanation :answer: Na and h which of the following atoms has the highest first ionization energy? A) h and br explanation: When there is less difference in electronegative of two atoms ( electronegativities are not same) difference in theirsize, polar covalent bond form. Web polar covalent bonds. For example, potassium nitrate, kno 3, contains the k + cation and the polyatomic no 3− anion. A) hand br ob) hand h c) na and br od) n and n.
Group 6a form 2 bonds; Web compounds with polar covalent bonds have electrons that are shared unequally between the bonded atoms. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Ionic bonds can be considered the ultimate in polarity, with electrons being transferred rather than shared. This problem has been solved!
Option (a) h and br. Web the combination of atoms that can form a polar covalent bond is h (hydrogen) and br (bromine). H and br explanation :answer: Advanced physics questions and answers. Two fluorine atoms, for example, can form a stable f 2 molecule in which each atom has an octet of valence electrons by sharing a pair of electrons. Only h and br form polar covalent bond.
Atoms will covalently bond with other atoms in order to gain more stability, which is gained by forming a full electron shell. Only h and br form polar covalent bond. Will form either one, two, three or four covalent bonds. Which combination of atoms can form a polar covalent bond? H and br explanation :answer:
In a polar covalent bond, sometimes simply called a polar bond,. In pure covalent bonds, the electrons are shared equally. Web covalent bonds are also affected by the electronegativity of the connected atoms which determines the chemical polarity of the bond. Ionic bonds can be considered the ultimate in polarity, with electrons being transferred rather than shared.
Web Which Combination Of Atoms Can Form A Polar Covalent Bond?
This problem has been solved! In polar covalent bonds, the electrons are shared unequally, as one atom exerts a stronger force of attraction on the electrons than the other. Option (a) h and br. Web the combination of atoms that can form a polar covalent bond is h (hydrogen) and br (bromine).
And Group 7A Form One Bond.
Web covalent bonds are also affected by the electronegativity of the connected atoms which determines the chemical polarity of the bond. Web polar covalent bonds. Web typically, the atoms of group 4a form 4 covalent bonds; This is due to very high electronegativity of the fluorine as a result it pulls electrons strongly.
However, These Polyatomic Ions Form Ionic Compounds By Combining With Ions Of Opposite Charge.
Web a polar covalent bond exists when atoms with different electronegativities share electrons in a covalent bond. Advanced physics questions and answers. Web a polar covalent bond is a bond formed when a shared pair of electrons are not shared equally. You'll get a detailed solution from a subject matter expert that helps you learn core concepts.
Na And H Which Of The Following Atoms Has The Highest First Ionization Energy?
For example, potassium nitrate, kno 3, contains the k + cation and the polyatomic no 3− anion. Group 6a form 2 bonds; When there is less difference in electronegative of two atoms ( electronegativities are not same) difference in theirsize, polar covalent bond form. An unequal relationship creates a polar covalent bond such as with h−cl.