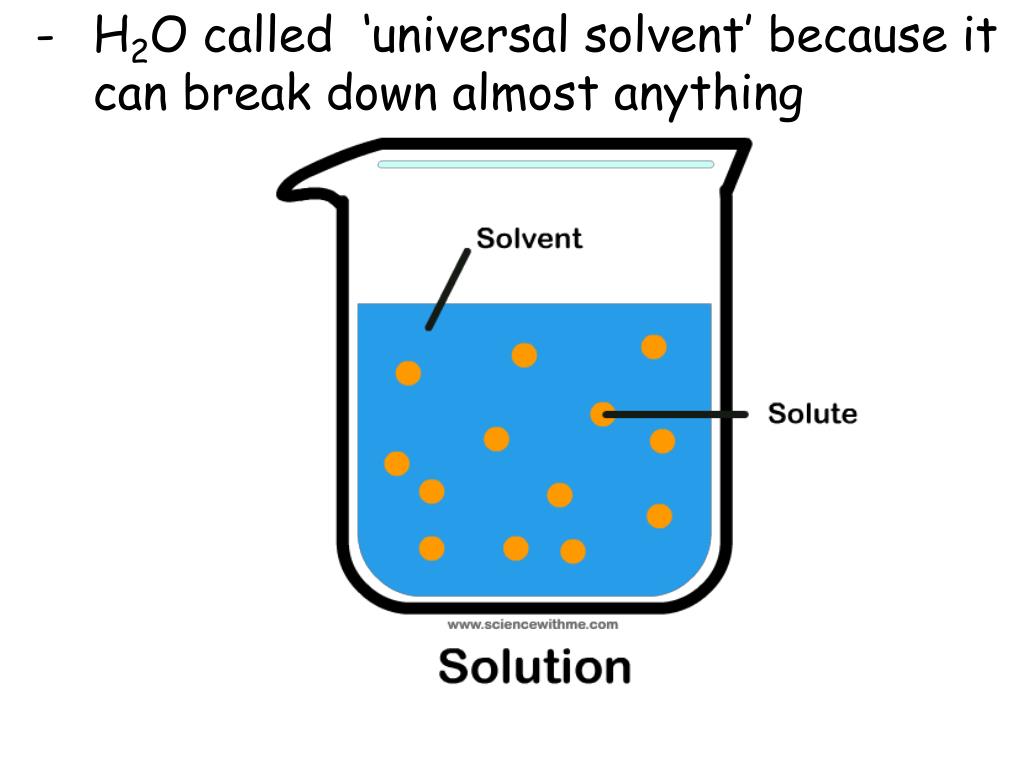
Universal Solvent Drawing
Universal Solvent Drawing - Use this animation to review this important vocabulary, solutes, solvents, and solutions. Web water is commonly referred to as the universal solvent, but what does that mean? Web thanks to its ability to dissolve a wide range of solutes, water is sometimes called the universal solvent. however, this name isn't entirely accurate, since there are some substances (such as oils) that don't dissolve well in water. Water sticks to pine needles due to adhesion; Here is an explanation of why water is called the universal solvent and what properties make it good at dissolving other substances. There are many individual elements and compounds that form ions. Are nonpolar water 'loving' molecules. Web water dissolves everything because it is the “universal solvent. water has the ability to dissolve many substances but the term “universal solvent is misleading. Here is a look at whether this is true and why water is so good at dissolving other compounds. Web water is the universal solvent because it is a polar molecule.
Water fountains work because of water's incompressibility; Chemistry makes water a great solvent. Solvents dissolve other substances, solutes dissolve in a solvent. A solvent is simply a liquid that other substances can dissolve in, and the reason that water has gained the label of universal solvent is because no other solvent can dissolve as many substances as it can. Water is capable of dissolving a variety of different substances, which is why it is such a good solvent. Web easily dissolve in water. Web view media details.
Web easily dissolve in water. Look for more clues & answers. Web water as the universal solvent because of water's polarity, it is able to dissolve or dissociate many particles. Cations, such as na (sodium) have a net positive charge, whereas anions (such as cl, chloride) have a net negative charge. Water fountains work because of water's incompressibility;
Web easily dissolve in water. Here is a look at whether this is true and why water is so good at dissolving other compounds. A universal solvent theoretically dissolves any other chemical. Web water is commonly referred to as the universal solvent, but what does that mean? Web thanks to its ability to dissolve a wide range of solutes, water is sometimes called the universal solvent. however, this name isn't entirely accurate, since there are some substances (such as oils) that don't dissolve well in water. Water is capable of dissolving a variety of different substances, which is why it is such a good solvent.
There are many individual elements and compounds that form ions. Web water as the universal solvent because of water's polarity, it is able to dissolve or dissociate many particles. Use this animation to review this important vocabulary, solutes, solvents, and solutions. Water is capable of dissolving a variety of different substances, which is why it is such a good solvent. Water sticks to pine needles due to adhesion;
This is important to every living thing on earth. It’s not actually universal, though, because it doesn’t dissolve nonpolar molecules very well. Web thanks to its ability to dissolve a wide range of solutes, water is sometimes called the universal solvent. however, this name isn't entirely accurate, since there are some substances (such as oils) that don't dissolve well in water. Water fountains work because of water's incompressibility;
Oxygen Has A Slightly Negative Charge, While The Two Hydrogens Have A Slightly Positive Charge.
Cations, such as na (sodium) have a net positive charge, whereas anions (such as cl, chloride) have a net negative charge. There are 3 different forms of water, or h 2 o: Web universal solvent definition. Generally, solvents dissolve similar things.
Web Water Dissolves Everything Because It Is The “Universal Solvent. Water Has The Ability To Dissolve Many Substances But The Term “Universal Solvent Is Misleading.
Here is a look at whether this is true and why water is so good at dissolving other compounds. We need to take the statement water is the universal solvent with a grain of salt (pun intended). Draw it draw the hydration shells that form around a potassium ion and a chloride ion when potassium chloride (kcl) dissolves in water. Web water is the universal solvent because it is a polar molecule.
Web Thanks To Its Ability To Dissolve A Wide Range Of Solutes, Water Is Sometimes Called The Universal Solvent. However, This Name Isn't Entirely Accurate, Since There Are Some Substances (Such As Oils) That Don't Dissolve Well In Water.
You may hear water called the universal solvent. Use this animation to review this important vocabulary, solutes, solvents, and solutions. Why water can look blue Water fountains work because of water's incompressibility;
Web Trish Gant/Getty Images.
Draw it draw the hydration shells that form around a potassium ion and a chloride ion when potassium chloride (kcl) dissolves in water. Label the positive, negative, and partial charges on the atoms. Water is known as the universal solvent. And, water is called the universal solvent because it dissolves more substances than any other liquid.