Isotopes Worksheet With Answers
Isotopes Worksheet With Answers - \textcolor {f21cc2} {\text {number of neutrons}=\text {mass number. Great practice for students to master atomic and mass numbers. How many protons and neutrons are in the first isotope? Answer the questions based on the above reading. These isotopes are neutral (charge = 0). 32 protons, 38 neutrons, 32 electrons. Web in a sample of e there are two isotopes. (i) 4019 p (ii) 12050 sn (iii) 2963 cu (iv) 10947 au (v) 5826 fe. For example, the atomic mass of carbon is reported as 12.011 amu (atomic mass units). Web ask the class to read through the remaining sections on the provided worksheet, and answer the questions.
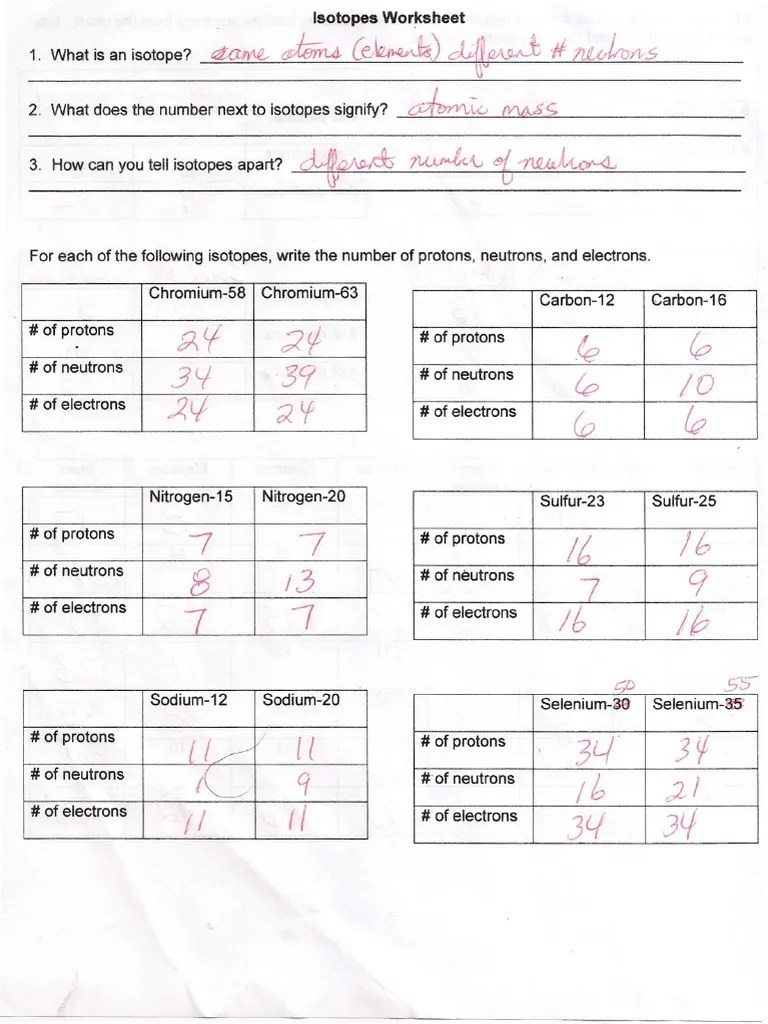
Give a reason for your answer. _____ _____ for each of the following isotopes, write the # of protons, neutrons, and electrons. Web are all atoms of an element the same? Here are three isotopes of an element: Web isotopes aqa gcse. 32 protons, 38 neutrons, 32 electrons. Web for each isotope shown, give the number of protons, neutrons, and electrons.
The number 6 refers to the _____ c. For example, the atomic mass of carbon is reported as 12.011 amu (atomic mass units). Great practice for students to master atomic and mass numbers. Web worksheets for a lesson on isotopes, including a full set of answers on powerpoint slides. Explain, in terms of subatomic particles, what is meant by the term isotopes.
Web atoms of the same element with different numbers of neutrons are called isotopes. Web in a sample of e there are two isotopes. Describe the general arrangement of subatomic particles in the atom electrons surround the nucleus; Fill in the following table. The lesson on “atomic structure and notation” is provided free in my other resources, so you can try before you buy. The numbers 12, 13, and 14 refer to the mass number.
The relative atomic mass (ar) of atoms is the average mass of all the different isotopes of an element (taking into account the amount of each isotope) on a scale where 12c atoms have a mass of exactly 12. Web for each isotope shown, give the number of protons, neutrons, and electrons. (i) 4019 p (ii) 12050 sn (iii) 2963 cu (iv) 10947 au (v) 5826 fe. _____ _____ for each of the following isotopes, write the # of protons, neutrons, and electrons. \textcolor {f21cc2} {\text {number of neutrons}=\text {mass number.
5 protons, 6 neutrons, 5 electrons. Web isotopes work sheet. Web are all atoms of an element the same? The number 6 refers to the _____ c.
The Relative Atomic Mass (Ar) Of Atoms Is The Average Mass Of All The Different Isotopes Of An Element (Taking Into Account The Amount Of Each Isotope) On A Scale Where 12C Atoms Have A Mass Of Exactly 12.
How many protons and neutrons are in the second isotope? \textcolor {f21cc2} {\text {number of neutrons}=\text {mass number. _____ _____ for each of the following isotopes, write the # of protons, neutrons, and electrons. Answer the questions based on the above reading.
Web Are All Atoms Of An Element The Same?
Identify which among the isotope symbols below is incorrect. Exshare answers from the class, ensuring that all students are able to press 18o/16o in terms of voltage and resistance before allowing them to move on to calculate 18o/16o ratio from the data provided. These isotopes are neutral (charge = 0). Great practice for students to master atomic and mass numbers.
Full Lesson For Gcse Chemistry With Worksheets On The Lesson Isotopes And Ions.
Which isotope of lead is likely to be the most abundant? The numbers 12, 13, and 14 refer to the mass number. 5 protons, 6 neutrons, 5 electrons. Describe the general arrangement of subatomic particles in the atom electrons surround the nucleus;
How Many Protons And Neutrons Are In The First Isotope?
What does the number next to isotopes signify? Explain, in terms of subatomic particles, what is meant by the term isotopes. The number 6 refers to the atomic number. Here are three isotopes of an element: