Isotopes Worksheet Answer Key
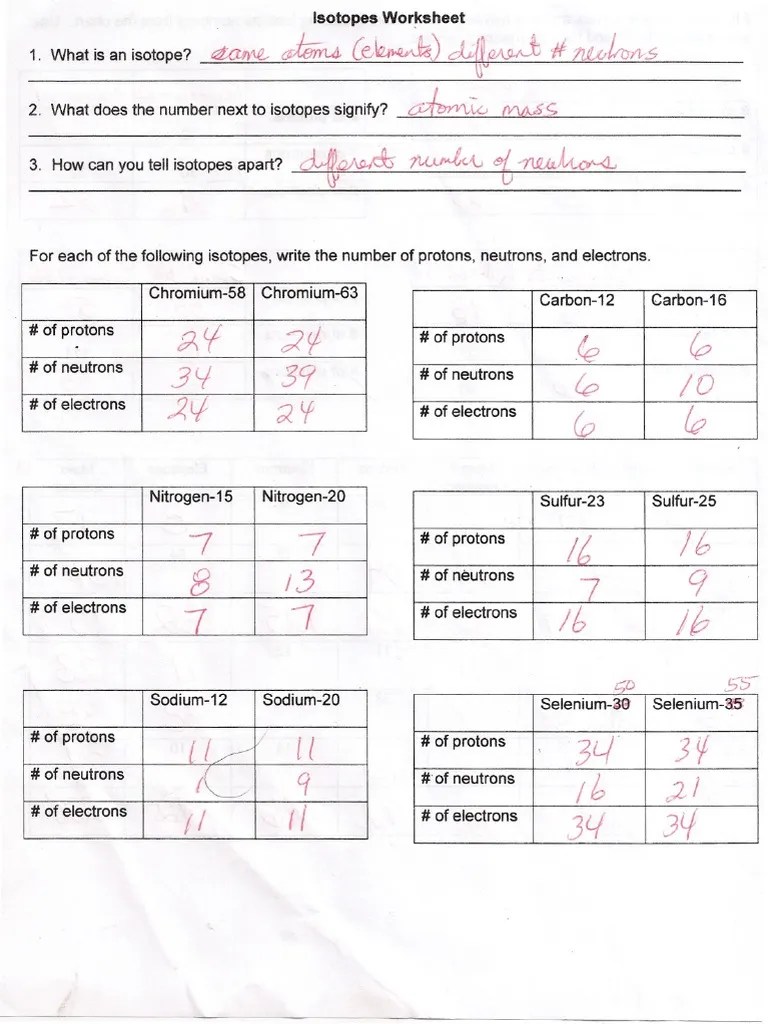
Isotopes Worksheet Answer Key - The number 6 refers to the atomic number. The number 6 refers to the atomic number c. 6 12c 6 13c 6 14c a. For example, the atomic mass of carbon is reported as 12.011 amu (atomic mass units). The number 6 refers to the _________________________ c. Web for each isotope shown, give the number of protons, neutrons, and electrons. Protons and neutrons are in the nucleus. Isotopes are versions of the same element. Answer the questions based on the above reading. Ask the class to read through the remaining sections on the provided worksheet, and answer the questions.
Isotopes are versions of the same element. 5 protons, 6 neutrons, 5 electrons. Web isotope practice worksheet 1. Students shared 4222 documents in this course. Web isotopes worksheet answer key. 32 protons, 38 neutrons, 32 electrons. Here are three isotopes of an element:
Are all atoms of an element the same? 6 12c 6 13c 6 14c a. Therefore ,they are the same element, but with different masses. A short worksheet to introduce or revise isotopes. Web isotopes of an element have the same atomic number (z) but have different mass numbers (a), because they have different numbers of neutrons.
Isotopes, isotope notation, neutrons, atomic mass. Therefore ,they are the same element, but with different masses. Web use the sim to learn about isotopes and how abundance relates to the average atomic mass of an element. Web this worksheet contains basic conceptual questions about atomic number, mass number, and isotopes. Fill in the isotope names and any missing information, including isotope numbers from the chart. Web isotope practice worksheet 1.
Fill in the isotope names and any missing information, including isotope numbers from the chart. Here are three isotopes of an element: Use your periodic table and the information provided. Here are three isotopes of an element: How can you tell one isotope from another?
Great practice for students to master atomic and mass numbers. Calculate the average atomic mass of chlorine if its isotopes and % abundances are as follows. Fill in the following table. 5 protons, 6 neutrons, 5 electrons.
Web Isotopes Of An Element Have The Same Atomic Number (Z) But Have Different Mass Numbers (A), Because They Have Different Numbers Of Neutrons.
How many protons and neutrons are in the first. The lesson on “atomic structure and notation” is provided free in my other. Atoms with same atomic number (same protons),but different # of neutrons. They have the same number of protons and electrons as the element but different mass numbers and number of neutrons.
Web Use The Sim To Learn About Isotopes And How Abundance Relates To The Average Atomic Mass Of An Element.
Protons and neutrons are in the nucleus. Web for each isotope shown, give the number of protons, neutrons, and electrons. Web fill in the isotope names and any missing information, including isotope numbers from the chart. How many protons and neutrons are in the first isotope?
How Many Protons And Neutrons Are In The First Isotope?
Answer the questions based on the above reading. The number indicates the isotope’s mass number. 6 12c 6 13c 6 14c a. The numbers 12, 13, and 14 refer to the ________________________ d.
Printable And Editable Student Worksheet (Pdf And Word Document) Paperless Digital Version For Use In Google Drive (Prepared With Google Slides) Complete Answer Key.
To find the most naturally occurring mass, you should round the atomic mass from the periodic table to the nearest whole number. What does the number next to isotopes signify? The numbers 12, 13, and 14 refer to the mass number d. It is often assumed that by key stage 5, students have a strong understanding.