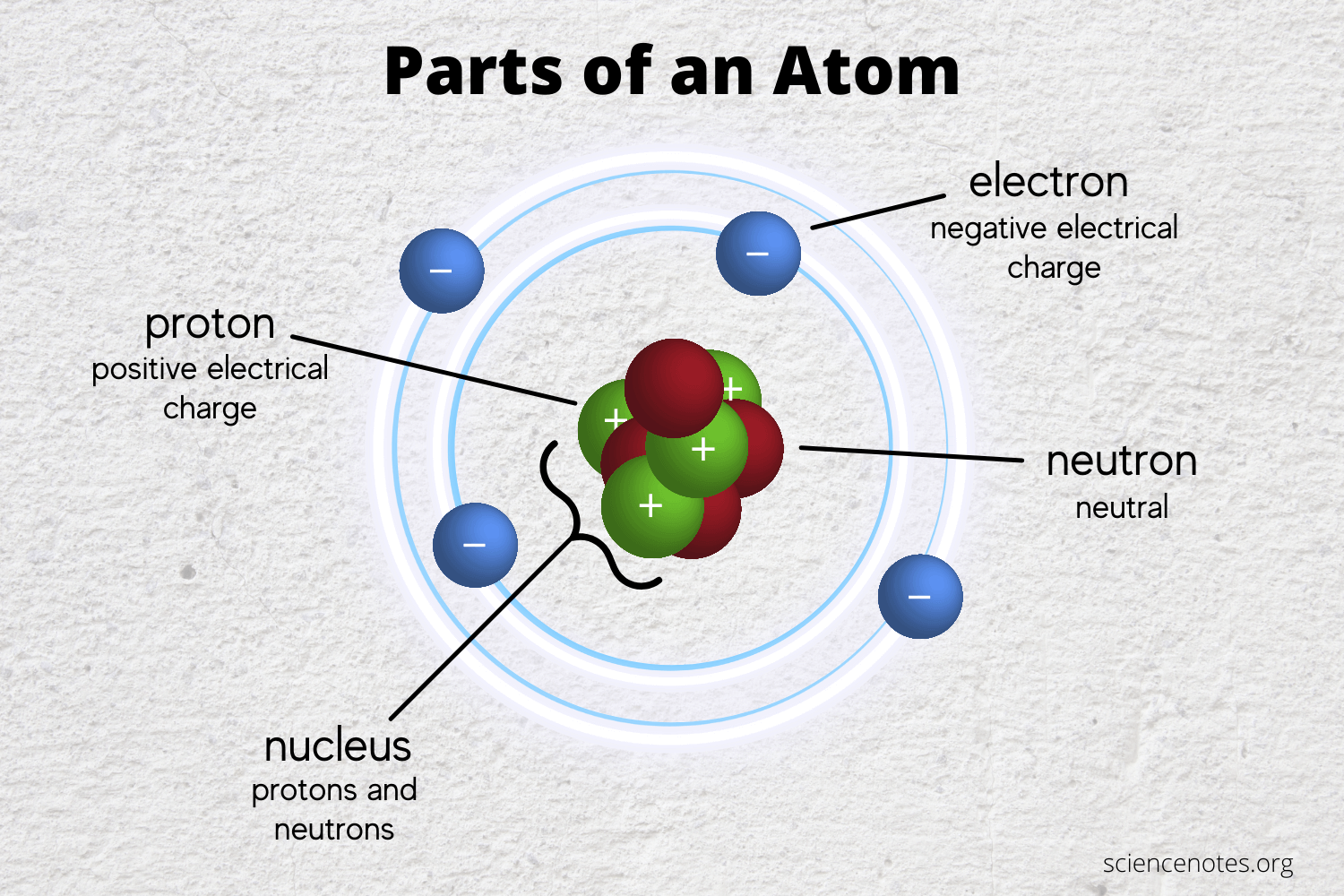
In General What Determines Whether Atoms Will Form Chemical Bonds
In General What Determines Whether Atoms Will Form Chemical Bonds - Web chemical bonds form when electrons can be simultaneously close to two or more nuclei, but beyond this, there is no simple, easily understood theory that would not. Learn about ionic bonds, covalent bonds, polyatomic ions, and metallic bonds, and how they lead to the. Web in general what determines whether atoms will form chemical bonds? A molecule is a neutral group of atoms that are held together by covalent bonds. The bond may result from the electrostatic force between oppositely. Web why do some atoms join together to form molecules, but others do not? Web in general, what determines whether atoms will form chemical bonds? Chemical bonds are formed when electrons in different atoms. Web when atoms bond together, they create molecules: Typically they join together in such a way that they lose their identities as elements and adopt a new identity as a compound.
Typically they join together in such a way that they lose their identities as elements and adopt a new identity as a compound. Web discover how atoms and ions come together through chemical bonding! A sodium atom bonds with a chlorine atom to create salt (sodium chloride), two hydrogen atoms bond with an. A chemical bond is a mutual electrical attraction between the nuclei in valence, electrons of different atoms that binds the atoms. Web the number and arrangement of electrons of an atom determine the kinds of chemical bonds that it forms and how it reacts with other atoms to form molecules. Web determine whether each of the following bonds would be polar or nonpolar: Learn about ionic bonds, covalent bonds, polyatomic ions, and metallic bonds, and how they lead to the.
Web in general what determines whether atoms will form chemical bonds? Web atoms can join together by forming a chemical bond, which is a very strong attraction between two atoms. (1) covalent bonding, in which. In order for a sodium atom to lose an electron, it needs to. Web in general, the loss of an electron by one atom and gain of an electron by another atom must happen at the same time:
Web in general what determines whether atoms will form chemical bonds? Web in general, the loss of an electron by one atom and gain of an electron by another atom must happen at the same time: Chemical bonds are formed when electrons in different atoms. Web the number and arrangement of electrons of an atom determine the kinds of chemical bonds that it forms and how it reacts with other atoms to form molecules. Web a mutual electrical attraction between the nuclei and valance electrons of different atoms that binds the atoms together. Web a chemical bond is the force that holds atoms together in chemical compounds.
The bond is caused by the. Why is the co 2 molecule linear whereas h 2 o is bent? Web the shell closest to the nucleus, 1n, can hold two electrons, while the next shell, 2n, can hold eight, and the third shell, 3n, can hold up to eighteen. Web atoms can join together by forming a chemical bond, which is a very strong attraction between two atoms. Chemical bonds are formed when electrons in different atoms.
Web the number and arrangement of electrons of an atom determine the kinds of chemical bonds that it forms and how it reacts with other atoms to form molecules. Web a chemical bond is the association of atoms or ions to form molecules, crystals, and other structures. In order for a sodium atom to lose an electron, it needs to. (1) covalent bonding, in which.
Web The Shell Closest To The Nucleus, 1N, Can Hold Two Electrons, While The Next Shell, 2N, Can Hold Eight, And The Third Shell, 3N, Can Hold Up To Eighteen.
Web why do some atoms join together to form molecules, but others do not? Typically they join together in such a way that they lose their identities as elements and adopt a new identity as a compound. The bond may result from the electrostatic force between oppositely. Web discover how atoms and ions come together through chemical bonding!
The Bond Is Caused By The.
Web determine whether each of the following bonds would be polar or nonpolar: Web in general, the loss of an electron by one atom and gain of an electron by another atom must happen at the same time: Web when atoms bond together, they create molecules: Web a mutual electrical attraction between the nuclei and valance electrons of different atoms that binds the atoms together.
Web A Chemical Bond Is An Attraction Between Atoms That Allows The Formation Of Chemical Substances That Contain Two Or More Atoms.
Web a chemical bond is the association of atoms or ions to form molecules, crystals, and other structures. A chemical bond is a mutual electrical attraction between the nuclei in valence, electrons of different atoms that binds the atoms. Web atoms can join together by forming a chemical bond, which is a very strong attraction between two atoms. Web in general, what determines whether atoms will form chemical bonds?
Web The Number And Arrangement Of Electrons Of An Atom Determine The Kinds Of Chemical Bonds That It Forms And How It Reacts With Other Atoms To Form Molecules.
A molecule is a neutral group of atoms that are held together by covalent bonds. Why is the co 2 molecule linear whereas h 2 o is bent? Web in general what determines whether atoms will form chemical bonds? What determines whether atoms will form chemical bonds is to maximize the stability.