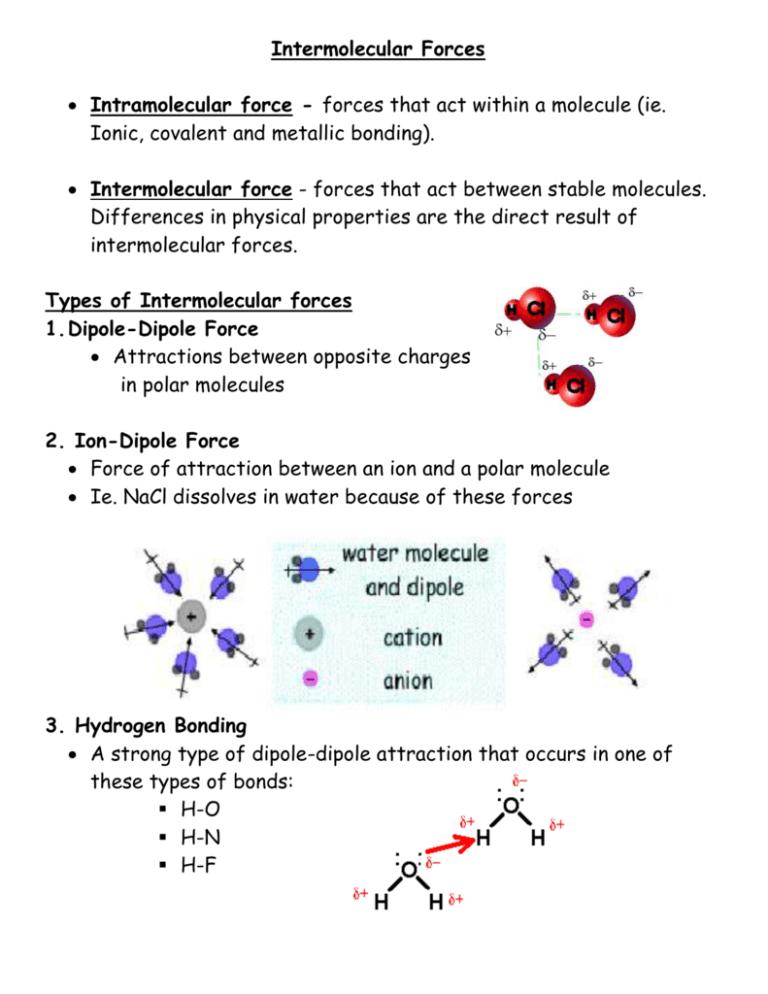
Imf Intermolecular Forces Worksheet
Imf Intermolecular Forces Worksheet - Which compound in each pair below would be expected to have the higher boiling point? List all of the intermolecular forces that are present for each of the following: Explain how imfs impact boiling points. Then in the last column, indicate which member of the pair you would expect to have the higher boiling point. Bonding & imf worksheets and answer keys. Many students confuse imfs with intramolecular forces, which were the center of the last unit. List the four major types of imfs. Question answer 1 define the terms: Explain how imfs impact melting points. Predict whether there is an overall molecular dipole.
Web this document provides a worksheet on intermolecular forces. A electronegativity b permanent dipole c polar molecule. Web intermolecular forces worksheet #1. Intermolecular forces (imf) can be qualitatively ranked using coulomb's law: Nonpolar molecules & their properties. Web understand the concepts of critical pressure, critical temperature, vapor pressure, normal boiling point, normal melting point, critical point, and triple point. Substance #1 dominant intermolecular force substance #2 dominant.
It contains questions about identifying imfs in molecules based on their structure, ranking imf strengths between substances, and ordering compounds by relative boiling points based on imf interactions. Understand the roles of enthalpy and entropy in determining solubility. Web since surface tension and viscosity are both greater with higher intermolecular forces the same trend will be seen where these bulk properties will lower with the decreased amount of intermolecular forces and increased temperature. Web determine the dominant intermolecular forces (imfs) of organic compounds. List all of the intermolecular forces that are present for each of the following:
Web since surface tension and viscosity are both greater with higher intermolecular forces the same trend will be seen where these bulk properties will lower with the decreased amount of intermolecular forces and increased temperature. Then in the last column, indicate which member of the pair you would expect to have the higher boiling point. Web this discussion worksheet addresses intermolecular forces separated into four categories: Web intermolecular forces worksheet #1. The forces that hold molecules together in the liquid and solid states are called. Then indicate what type of bonding is holding the atoms together inone molecule of the following.
Chemical bonds are intramolecular forces which hold atoms together as molecules. List all of the intermolecular forces that are present for each of the following: Try to remember the following: Draw lewis structures for these molecules. Indicate thestrongest imf holding togetherthousands of molecules of the following.
Understand the roles of enthalpy and entropy in determining solubility. What other factor affects melting points? The properties of liquids are intermediate between those of gases and solids, but are more similar to solids. Click types of imf.doc link to view the file.
Web Intermolecular Forces (Imfs) Are The Attractive Or Repulsive Forces Between Entire Molecules Due To Differences In Charge.
Draw lewis structures for these molecules. Web the forces that hold molecules together in the liquid and solid states are called intermolecular forces and are appreciably weaker. List all types of imfs that would occur in each of the following (you should have a good. Which compound in each pair below would be expected to have the higher boiling point?
Which Gas Would Be Expected To Have A Greater Boiling Point:
Imf and impurities in melting points; Predict whether there is an overall molecular dipole. Web since surface tension and viscosity are both greater with higher intermolecular forces the same trend will be seen where these bulk properties will lower with the decreased amount of intermolecular forces and increased temperature. Then indicate what type of bonding is holding the atoms together inone molecule of the following.
Web Determine The Dominant Intermolecular Forces (Imfs) Of Organic Compounds.
Predict the molecular shape of each of the following: List the four major types of imfs. Intermolecular forces (imf) can be qualitatively ranked using coulomb's law: What other factor affects melting points?
It Contains Questions About Identifying Imfs In Molecules Based On Their Structure, Ranking Imf Strengths Between Substances, And Ordering Compounds By Relative Boiling Points Based On Imf Interactions.
Question answer 1 define the terms: Web this discussion worksheet addresses intermolecular forces separated into four categories: The properties of liquids are intermediate between those of gases and solids, but are more similar to solids. Explain how imfs impact boiling points.