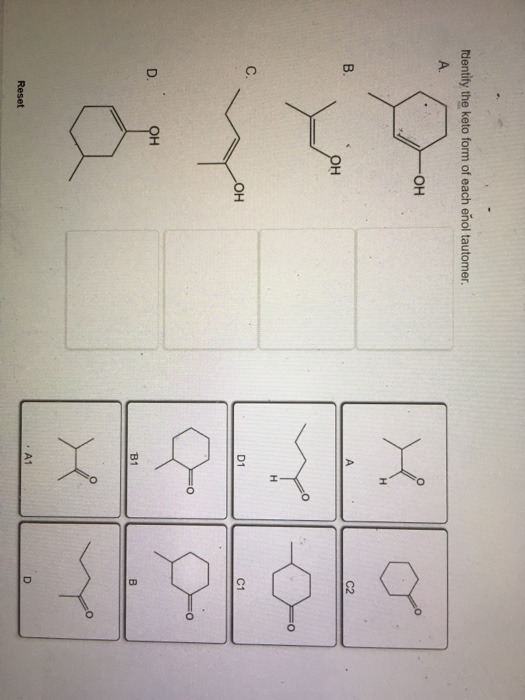
Identify The Keto Form Of Each Enol Tautomer
Identify The Keto Form Of Each Enol Tautomer - In this chapter on alpha carbon chemistry, you are going to come across the term “enol” almost in any new reaction. The chemical equilibrium is thermodynamically driven. Topics covered in other articles. Identify the keto form of each enol tautomer. One applies to acidic conditions, while the other applies to basic conditions. Web the individual keto and enol isomers are called tautomers. Web in a solution, you won't see much of the enol form, but these can occur. You will use nmr spectroscopy to determine the equilibrium constant for the reaction, and then investigate the reasons for any differences in the equilibrium constants via computational methods. There are two distinct reaction pathways to be considered since the presence of either acidic or basic conditions leads to tautomerism. Enol tautomer → keto tautomer.
For alkylation reactions of enolate anions to be useful, these intermediates must be generated in high concentration in the absence of other strong nucleophiles and bases. Make certain that you can define, and use in context, the key terms below. There are two distinct reaction pathways to be considered since the presence of either acidic or basic conditions leads to tautomerism. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. In this chapter on alpha carbon chemistry, you are going to come across the term “enol” almost in any new reaction. Then lone pair electrons from the enolate anion attack an electrophilic h + through conjugation with the double bond. Web in a solution, you won't see much of the enol form, but these can occur.
And so you could imagine, these are tautomers, so this mechanism is actually called a tautomerization, and these are the keto and enol forms of the tautomers. Of the spectral line width. View the full answer step 2. This problem has been solved! The oxonium ion undergoes resonance stabilization by moving the π electrons from the c=o bond towards the protonated oxygen.
Web thus, tautomerism describes the equilibrium between keto and enol forms interconverted through a change in the position of bonding electrons and hydrogen to produce two isomers. Make certain that you can define, and use in context, the key terms below. Note the difference between tautomers and resonance forms. Ldentify the enol form of each keto tautomer a. Identify the keto form of each enol tautomer. You will use nmr spectroscopy to determine the equilibrium constant for the reaction, and then investigate the reasons for any differences in the equilibrium constants via computational methods.
Web in a solution, you won't see much of the enol form, but these can occur. Tautomers are constitutional isomers—different compounds with different structures—while resonance forms are different representations of a single compound. View the full answer step 2. Identify the keto form of each enol tautomer. Identify the keto form of each enol tautomer.
This problem has been solved! And so you could imagine, these are tautomers, so this mechanism is actually called a tautomerization, and these are the keto and enol forms of the tautomers. Web the keto and enol forms are known as tautomers and they constantly interconvert (or tautomerize) between the two forms under acid or base catalyzed conditions. In addn., no spectroscopic evidence for fast proton transfer from the enol form to the keto form in the d0 state has been obtained from the anal.
One Applies To Acidic Conditions, While The Other Applies To Basic Conditions.
Web the individual keto and enol isomers are called tautomers. Note the difference between tautomers and resonance forms. In this chapter on alpha carbon chemistry, you are going to come across the term “enol” almost in any new reaction. Identify the keto form of each enol tautomer.
Web In A Solution, You Won't See Much Of The Enol Form, But These Can Occur.
For alkylation reactions of enolate anions to be useful, these intermediates must be generated in high concentration in the absence of other strong nucleophiles and bases. You will use nmr spectroscopy to determine the equilibrium constant for the reaction, and then investigate the reasons for any differences in the equilibrium constants via computational methods. Then lone pair electrons from the enolate anion attack an electrophilic h + through conjugation with the double bond. Make certain that you can define, and use in context, the key terms below.
Web Thus, Tautomerism Describes The Equilibrium Between Keto And Enol Forms Interconverted Through A Change In The Position Of Bonding Electrons And Hydrogen To Produce Two Isomers.
Because carbonyl groups are sp 2 hybridized the carbon and oxygen both have un hybridized p orbitals which can overlap to form the c=o π π bond. Tautomers are constitutional isomers—different compounds with different structures—while resonance forms are different representations of a single compound. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. The α hydrogen is then deprotonated to form the enol tautomer.
Often, The Position Of Carbon Atoms Near A Carbonyl Group Are Designated By Greek Letters.
The atom adjacent to the carbonyl is alpha, the next removed is beta and so on. Enol tautomer → keto tautomer. Of the spectral line width. Web the individual keto and enol isomers are called tautomers.