How Does Pyrite Form
How Does Pyrite Form - Web pyrite, also known as “fool’s gold,” is a common iron sulfide mineral with the chemical formula fes2. Slower cooling results in larger crystals. Bisulphite can be partially oxidised or can react with organic matter and reactive metal species. Sulfur, which can occur in coal and is a pollutant when burned, occurs in three forms. The process of pyrite formation in sediments results from the action of bacteria, which reduce sulphate ions (dissolved in the pore water) to sulphide. These compounds were formed after igneous rocks are pressurized, heated, and subsequently changed over millions of years. Web pyrite, a naturally occurring iron disulfide mineral. Web it forms when iron and sulfur combine in specific geological conditions that typically involve the presence of water and oxygen. Web sedimentary pyrite formation during early diagenesis is a major process for controlling the oxygen level of the atmosphere and the sulfate concentration in seawater over geologic time. To the novice its colour is deceptively similar to that of a gold nugget.
Pyrite is called fool’s gold; It has a chemical composition of iron sulfide (fes 2) and is the most common sulfide mineral. Web pyrite, also known as “fool’s gold,” is a common iron sulfide mineral with the chemical formula fes2. Sulfur, which can occur in coal and is a pollutant when burned, occurs in three forms. Pyrite forms under reducing conditions, typically in environments with low oxygen levels, high sulfur content, and abundant iron. One of those forms is pyritic sulfur, which occurs as iron sulfide or. It forms in a variety of geological settings through several processes.
Pyrite forms under reducing conditions, typically in environments with low oxygen levels, high sulfur content, and abundant iron. Howstuffworks | aug 25, 2023. Iron sulfide, sometimes containing small amounts of cobalt, nickel, silver, and gold. Web how does pyrite form (pyritisation)? Web pyrite, a naturally occurring iron disulfide mineral.
Healing properties & everyday uses. The specimen’s size can vary. Web pyrite, a naturally occurring iron disulfide mineral. Web the relatively rapid formation of pyrite framboids explains how pyrite infills and preserves soft tissues before cell lysis and before deformation through burial has been initiated. One of those forms is pyritic sulfur, which occurs as iron sulfide or. Bacteria breaks down the organics forming bisulphide, the s2 component of pyrite.
These chlorite compounds contain the iron found in pyrite, but they also contain silicone and oxygen, which must be removed and. Sulfur, which can occur in coal and is a pollutant when burned, occurs in three forms. Web although pyrite is quite common worldwide, the disk form is virtually unique to illinois. It forms in a variety of geological settings through several processes. Web where it is found in igneous rocks, it may have been formed from magma in which the minerals were heated into a melted mass, and then the various minerals separated out at different temperatures, forming crystals as it cooled;
Web where it is found in igneous rocks, it may have been formed from magma in which the minerals were heated into a melted mass, and then the various minerals separated out at different temperatures, forming crystals as it cooled; It has a chemical composition of iron sulfide (fes 2) and is the most common sulfide mineral. Web pyrite, or iron disulfide, is the most common sulfide mineral on the earth’s surface and is widespread through the geological record. Web the formation of pyrite crystals depends mainly on the iron content of the sediment.
Web Pyrite, A Naturally Occurring Iron Disulfide Mineral.
The specimen’s size can vary. Bacteria breaks down the organics forming bisulphide, the s2 component of pyrite. Sulfur, which can occur in coal and is a pollutant when burned, occurs in three forms. The process of pyrite formation in sediments results from the action of bacteria, which reduce sulphate ions (dissolved in the pore water) to sulphide.
Web Sedimentary Pyrite Formation During Early Diagenesis Is A Major Process For Controlling The Oxygen Level Of The Atmosphere And The Sulfate Concentration In Seawater Over Geologic Time.
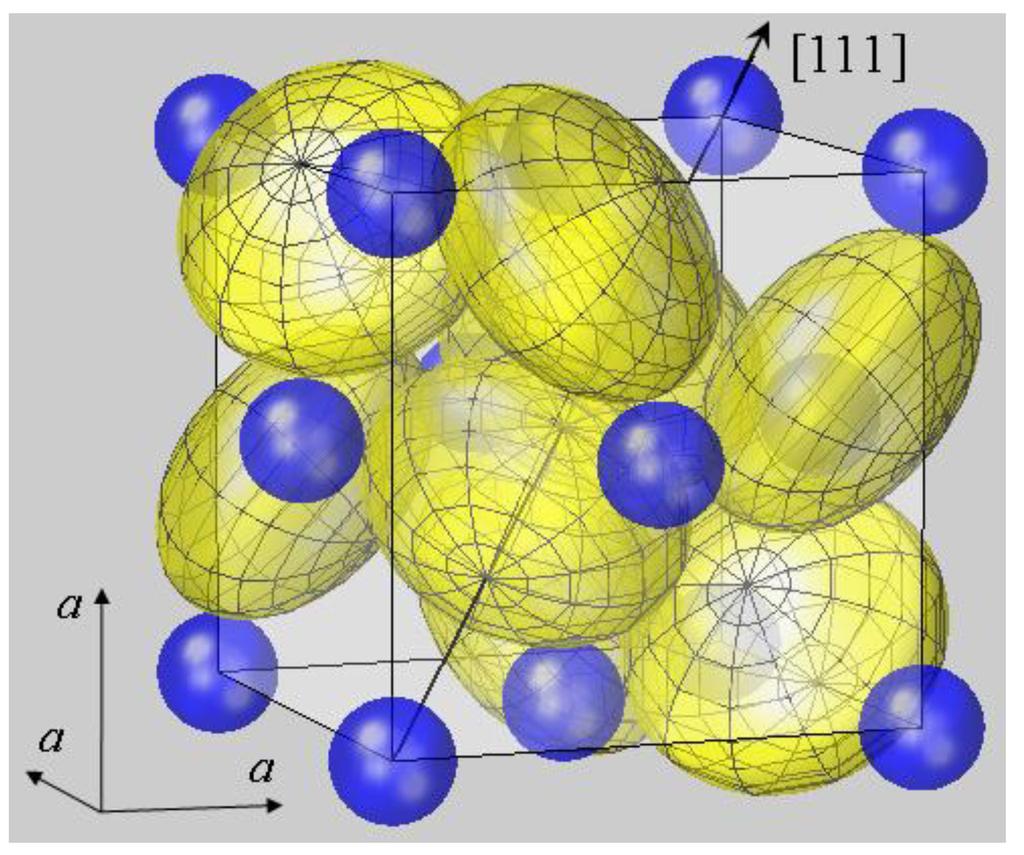
Bacterial sulphate reduction produces bisulphide. These chlorite compounds contain the iron found in pyrite, but they also contain silicone and oxygen, which must be removed and. One of those forms is pyritic sulfur, which occurs as iron sulfide or. Web pyrite ( fesx2 f e s x 2) forms cubic crystals, like these ones:
Iron Sulfide, Sometimes Containing Small Amounts Of Cobalt, Nickel, Silver, And Gold.
To the novice its colour is deceptively similar to that of a gold nugget. Web pyrite, or iron disulfide, is the most common sulfide mineral on the earth’s surface and is widespread through the geological record. Web the formation of pyrite crystals depends mainly on the iron content of the sediment. Pyrite forms under reducing conditions, typically in environments with low oxygen levels, high sulfur content, and abundant iron.
These Compounds Were Formed After Igneous Rocks Are Pressurized, Heated, And Subsequently Changed Over Millions Of Years.
It forms in a variety of geological settings through several processes. Web it forms when iron and sulfur combine in specific geological conditions that typically involve the presence of water and oxygen. Web the formation of pyrite starts with chlorite iron deposits within the ground. Over time, iron sulfide minerals crystallize, resulting in pyrite’s formation.