Glucose Generally E Ists In Ring Form
Glucose Generally E Ists In Ring Form - The rest is one of two cyclic forms of glucose formed when the hydroxyl group on carbon 5 (c 5) bonds to the aldehyde carbon 1 (c 1 ), as shown below. Web glucose generally exists in ring (cyclic) form. Glucose generally exists in ring (cyclic) form. Web glucose molecules form rings. Web the two anomers equilibrate in aqueous solution, a process known as mutarotation. Linear glucose has four chiral carbons (pointed to by the red arrows). The ring form is more stable, but in solution all three structures are present. Web glucose is usually present in solid form as a monohydrate with a closed pyran ring (dextrose hydrate). Web glucose ring form and straight line form when glucose forms the ring structure, it can form two isomers. This rotation produces either of.
When we prepare solid glucose, we usually get either the alpha or the beta form of the. A haworth projection shows the orientations of the hydroxyl groups and hydrogen atoms on the ring. Look at the orientation of the hydroxyl group at c2. These two forms always exist in equilibrium. Web glucose is usually present in solid form as a monohydrate with a closed pyran ring (dextrose hydrate). Web it is possible to obtain a sample of crystalline glucose in which all the molecules have the α structure or all have the β structure. Glucose generally exists in ring (cyclic) form.
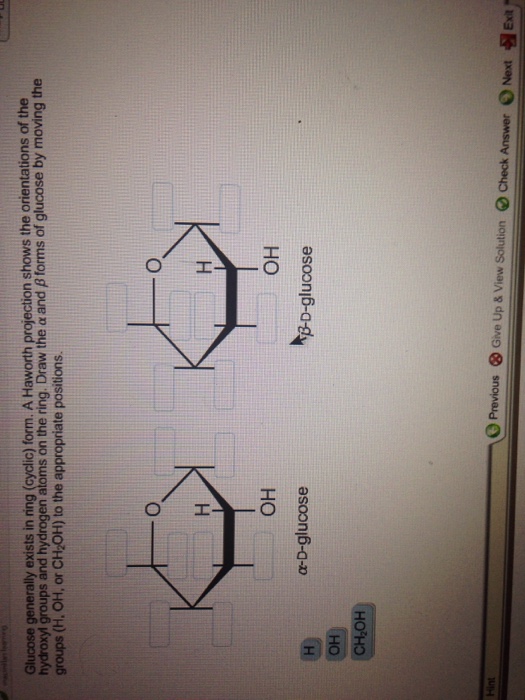
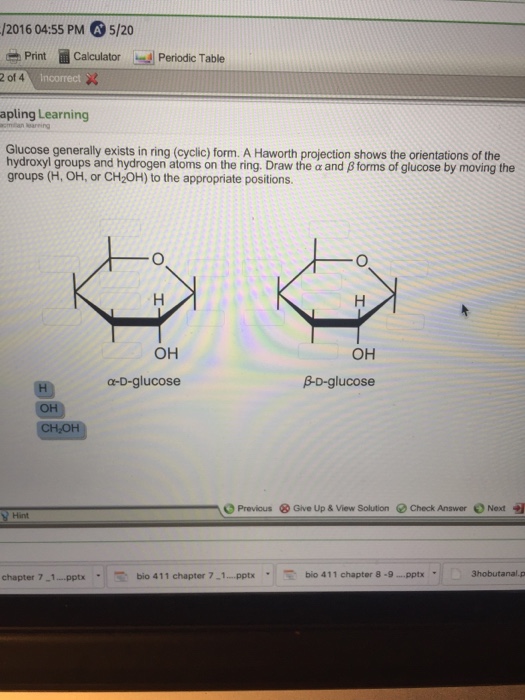
Forms of glucose by moving the groups (h, oh, or ch2oh) to the appropriate positions. Web it is possible to obtain a sample of crystalline glucose in which all the molecules have the α structure or all have the β structure. Whereas glucose and galactose are predominantly found in the β anomeric form, mannose is most frequently found as α anomer. When that happens, there are two possible orientations. Web sugars in aqueous solution exist in an equilibrium between the linear form and the ring form, which is formed by intramolecular attack by a hydroxyl group on the carbonyl.
Is the alpha or the beta form “really” glucose? Draw the \alpha α and \beta β forms of glucose by moving the groups (h, oh, or ch2oh) to the appropriate positions. Note that this rearrangement does not change the relative positions of hydroxyl groups. Web in aqueous solution, glucose exists in both the open and closed forms. A haworth projection shows the orientations of the hydroxyl groups and the hydrogen atoms on the ring. Well, all three molecules are forms of glucose.
Forms of glucose by moving the groups (h, oh, or ch2oh) to the appropriate positions. Draw the \alpha α and \beta β forms of glucose by moving the groups (h, oh, or ch2oh) to the appropriate positions. A haworth projection shows the orientations of the hydroxyl groups and hydrogen atoms on the ring. Web the most prevalent form for most carbohydrates is a ring. Web glucose generally exists in ring (cyclic) form.
Web it is possible to obtain a sample of crystalline glucose in which all the molecules have the α structure or all have the β structure. Web independent of stereoisomerization, sugars in ring form of a given type (such as glucose) can “twist themselves into alternative conformations called boat and chair. Web glucose is usually present in solid form as a monohydrate with a closed pyran ring (dextrose hydrate). The ring form is more stable, but in solution all three structures are present.
Web Glucose Is Usually Present In Solid Form As A Monohydrate With A Closed Pyran Ring (Dextrose Hydrate).
Web glucose ring form and straight line form when glucose forms the ring structure, it can form two isomers. The rest is one of two cyclic forms of glucose formed when the hydroxyl group on carbon 5 (c 5) bonds to the aldehyde carbon 1 (c 1 ), as shown below. Glucose generally exists in ring (cyclic) form. The ring form is more stable, but in solution all three structures are present.
Glucose Generally Exists In Ring (Cyclic) Form.
Draw the α and β forms of glucose by moving the groups (h, oh, or ch _2 2 oh) to the appropriate positions. Linear glucose has four chiral carbons (pointed to by the red arrows). Web the most prevalent form for most carbohydrates is a ring. A haworth projection shows the orientations of the hydroxyl groups and hydrogen atoms on the ring.
When That Happens, There Are Two Possible Orientations.
Web the two anomers equilibrate in aqueous solution, a process known as mutarotation. The process is catalyzed by acid, since hemiacetal formation is catalyzed by acid. A haworth projection shows the orientations of the hydroxyl groups and hydrogen atoms on the ring. A haworth projection shows the orientations of the hydroxyl groups and hydrogen atoms on the ring.
One Of The Oxygens Farther Along The Chain Can Reach Around And Bond To The Carbon In The C=O At The Head Of The Chain.
The α form melts at 146°c and has a specific rotation of +112°, while the β form melts at 150°c and has a specific rotation of +18.7°. Note that this rearrangement does not change the relative positions of hydroxyl groups. Draw the α and β forms of glucose by moving the groups (h,oh, or ch2oh) to the appropriate positions. This rotation produces either of.