Does Hydrogen Form Ionic Bonds
Does Hydrogen Form Ionic Bonds - Web atoms interact with each other through the formation of chemical bonds. Ionic bonds result from the attraction between. Such a bond is weaker than an. Web a hydrogen atom with one electron and a chlorine atom with 17 electrons. Although not as bare hx+ h x +, it can form ionic bonds in the form of hx− h x − ( hydride anion ). Web hydrogen ionic bonds are characterised by the hydrogen atom sharing an electron, forming neutral bonds, low bond energy indicating weak bonds, and acting as a poor. Electrovalency, electrovalent bond, heteropolar bond, polar bond. After bonding, the chlorine atom is now in contact with eight electrons in its outer shell, so it is. Covalent and ionic bonds are intramolecular forces. The oxygen atom of dmso form a hydrogen bond to the cation [emim] \(^+\).
Hx− h x − forms ionic. Such a bond is weaker than an. The oxygen atom of dmso form a hydrogen bond to the cation [emim] \(^+\). Web a water molecule consists of two hydrogen atoms bonded to an oxygen atom, and its overall structure is bent. Identify the key difference between ionic and covalent bonds. Web atoms interact with each other through the formation of chemical bonds. Ionic bonds form when one atom.
Sometimes in compounds such as hcl there is an strong ionic bond but in compounds like ch4 hydrogen. Identify the key difference between ionic and covalent bonds. After bonding, the chlorine atom is now in contact with eight electrons in its outer shell, so it is. Ionic bonds form when one atom. Web the rdfs of the individual hydrogen bonds were averaged over all three ring protons.
Web hydrogen ionic bonds are characterised by the hydrogen atom sharing an electron, forming neutral bonds, low bond energy indicating weak bonds, and acting as a poor. Although not as bare hx+ h x +, it can form ionic bonds in the form of hx− h x − ( hydride anion ). Web hydrogen does form ionic bonds. Ionic bonds form when one atom. Hydrogen bonds are intermolecular forces; Covalent and ionic bonds are intramolecular forces.
After bonding, the chlorine atom is now in contact with eight electrons in its outer shell, so it is. Covalent and ionic bonds are intramolecular forces. Web hydrogen ionic bonds are characterised by the hydrogen atom sharing an electron, forming neutral bonds, low bond energy indicating weak bonds, and acting as a poor. Web science & tech. Ionic bonds form when one atom.
Although not as bare hx+ h x +, it can form ionic bonds in the form of hx− h x − ( hydride anion ). Web hydrogen bonding, interaction involving a hydrogen atom located between a pair of other atoms having a high affinity for electrons; Covalent and ionic bonds are intramolecular forces. Web a hydrogen atom with one electron and a chlorine atom with 17 electrons.
Web The Rdfs Of The Individual Hydrogen Bonds Were Averaged Over All Three Ring Protons.
Distinguish between nonpolar and polar covalent bonds. Hydrogen bonds are intermolecular forces; Web distinguish between ions, cations, and anions. Hx− h x − forms ionic.
They Are Not Ionic Bonds,.
Web atoms interact with each other through the formation of chemical bonds. One type of chemical bond is an ionic bond. A bond is ionic if the electronegativity difference between the atoms is great enough that one atom could pull an electron completely away from the other one. Web a water molecule consists of two hydrogen atoms bonded to an oxygen atom, and its overall structure is bent.
Web Hydrogen Bonds Are One Of The Principal Intermolecular Forces.
Electrovalency, electrovalent bond, heteropolar bond, polar bond. Identify the key difference between ionic and covalent bonds. Such a bond is weaker than an. After bonding, the chlorine atom is now in contact with eight electrons in its outer shell, so it is.
A Special Class Are Ionic Hydrogen Bonds (Ihbs) That Form Between Ions And Molecules With Bonds Strengths Of.
Web hydrogen bonding is the strongest type of intermolecular bond. Web as it turns out, the hydrogen is slightly negative. This is because the oxygen atom, in addition to forming bonds. The oxygen atom of dmso form a hydrogen bond to the cation [emim] \(^+\).

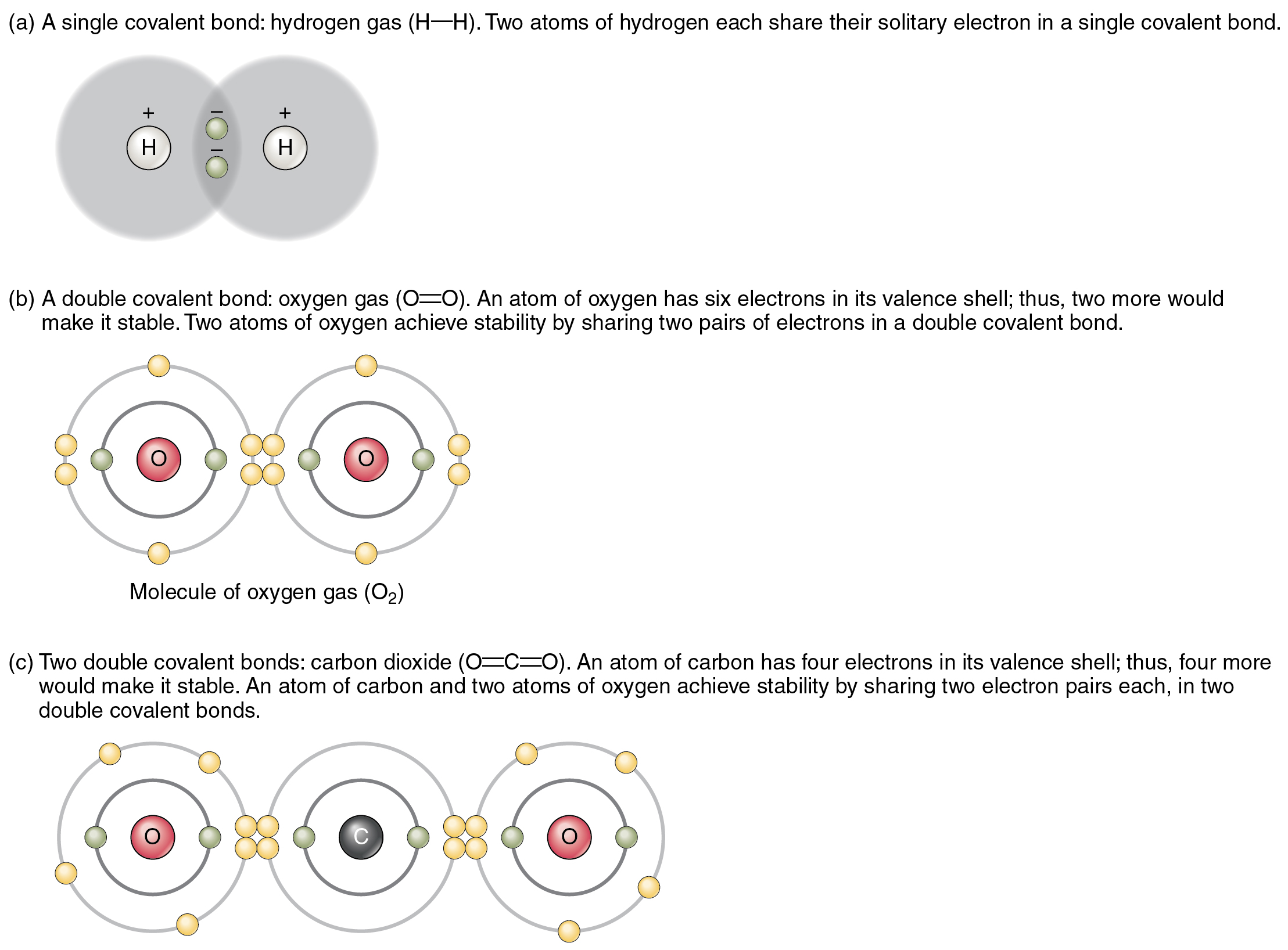



.PNG)
