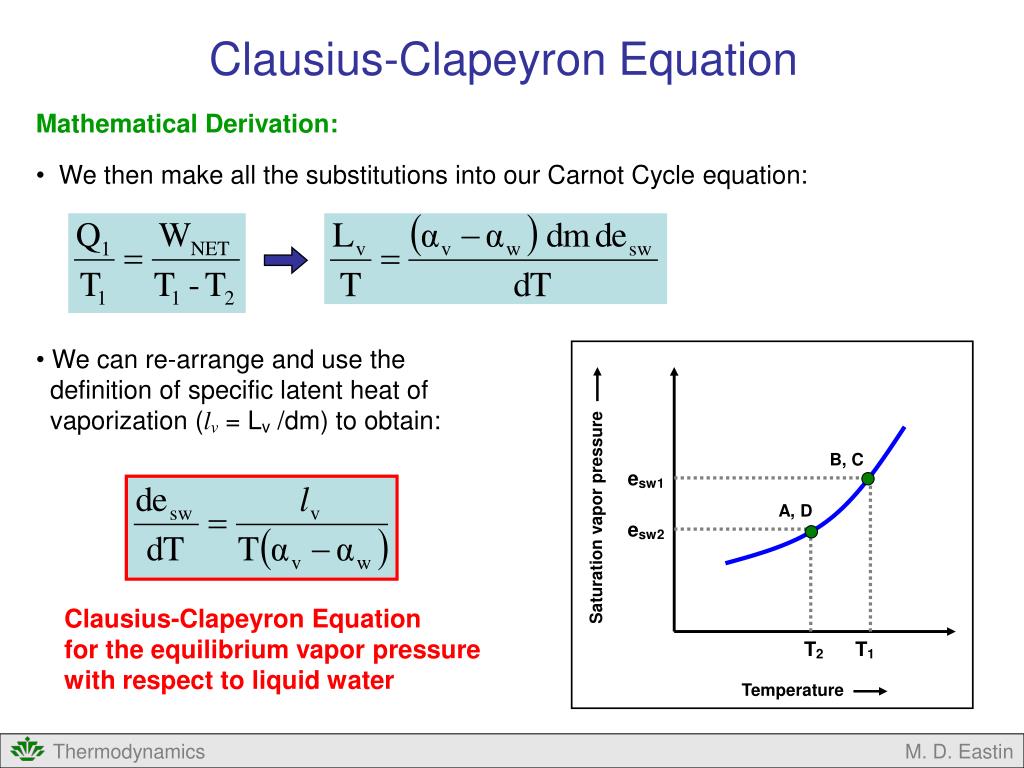
Clausius Clapeyron Equation E Ample
Clausius Clapeyron Equation E Ample - This equation is extremely useful in characterizing a discontinuous phase transition between two phases. Web to find the change in temperature, use the clapeyron equation (equation 8.4.4 8.4.4) and separating the variables. The clausius clapeyron equation predicts the rate at which vapour pressure increases per unit increase in temperature for a substance's vapour pressure (p) and temperature (t). At 100 o c the rate of increase of vapour pressure of steam is 27.1 mm hg per celsius degree, and a gram of steam occupies 1674 cm 3. Integration (with the assumption that δhfus/δv δ h f u s / δ v does not change much over the temperature range) yields. \frac {dp} {dt} = \frac {h} {t \cdot \delta v} dt dp = t ⋅ δv h. R is the ideal gas constant =. It is often used to calculate vapor pressure of a liquid. Web the clausius clapeyron equation includes: Δh vap is the enthalpy of vaporization of the solution.
\[\frac{d ln p}{dt} = \frac{\delta h_{vap}}{rt_{2}}\] To do so, the heat of vaporization and the specific volumes must be known functions of temperature. Dp = δhfus δv dt t d p = δ h f u s δ v d t t. This equation was suggested by b. It is often used to calculate vapor pressure of a liquid. Web t 1 = 287.85 k. 2) set up equation with values:
Clapeyron in 1834 and improved by r. Web t 1 = 287.85 k. Web to find the change in temperature, use the clapeyron equation (equation 8.4.4 8.4.4) and separating the variables. 509 applies to vaporization of liquids where vapor follows ideal gas law using the specific gas constant and liquid volume is neglected as being much smaller than vapor volume v. Web the clausius clapeyron equation includes:
Δh vap is the enthalpy of vaporization of the solution. It is often used to calculate vapor pressure of a liquid. Web using the values r = 8.3145 joules per k and λ = 40.65 kilojoules per mole, the above equation gives t = 342 k (69 °c) for the boiling temperature of water, which is barely enough to make tea. 2) set up equation with values: \frac {dp} {dt} dt dp. At 100 o c the rate of increase of vapour pressure of steam is 27.1 mm hg per celsius degree, and a gram of steam occupies 1674 cm 3.
Web the clausius clapeyron equation derivation. \frac {dp} {dt} dt dp. Clapeyron in 1834 and improved by r. This is the case for either sublimation ( solid → gas solid → gas) or vaporization ( liquid → gas liquid → gas ). At 100 o c the rate of increase of vapour pressure of steam is 27.1 mm hg per celsius degree, and a gram of steam occupies 1674 cm 3.
Clapeyron in 1834 and improved by r. Web using the values r = 8.3145 joules per k and λ = 40.65 kilojoules per mole, the above equation gives t = 342 k (69 °c) for the boiling temperature of water, which is barely enough to make tea. This is the case for either sublimation ( solid → gas solid → gas) or vaporization ( liquid → gas liquid → gas ). Web to find the change in temperature, use the clapeyron equation (equation 8.4.4 8.4.4) and separating the variables.
T 2 = 52.8 °C + 273.15.
Web the clausius clapeyron equation includes: Web the clapeyron equation can be developed further for phase equilibria involving the gas phase as one of the phases. Web how to calculate vapor pressure? This equation was suggested by b.
T 2 = 325.95 K.
Web the clausius clapeyron equation derivation. Dp = δhfus δv dt t d p = δ h f u s δ v d t t. This is the case for either sublimation ( solid → gas solid → gas) or vaporization ( liquid → gas liquid → gas ). — derivative of pressure with respect to.
Δh Vap Is The Enthalpy Of Vaporization Of The Solution.
2) set up equation with values: 509 applies to vaporization of liquids where vapor follows ideal gas law using the specific gas constant and liquid volume is neglected as being much smaller than vapor volume v. Let's have a closer look at two vapor pressure equations: We can further work our the integration and find the how the equilibrium vapor pressure changes with temperature:
\Frac {Dp} {Dt} Dt Dp.
This equation is extremely useful in characterizing a discontinuous phase transition between two phases. \[\frac{d ln p}{dt} = \frac{\delta h_{vap}}{rt_{2}}\] It is often used to calculate vapor pressure of a liquid. To do so, the heat of vaporization and the specific volumes must be known functions of temperature.