Can Serine Form Hydrogen Bonds
Can Serine Form Hydrogen Bonds - Hydrogenation of serine gives the diol serinol: This is a classic situation where hydrogen bonding can occur. Web thus, hydrogen bonds always contain three atoms and only those three. The opposite behavior is seen for cysm, as this residue seems to prefer more exposure than cys to the solvent. Tyrosine possesses a hydroxyl group in the aromatic ring, making it a phenol derivative. These attractions are an example of hydrogen bonds, weak interactions that form between a hydrogen with a partial positive charge and a more electronegative atom, such as oxygen. Answer only one, the one at the very top which is attached to the highly electrongative oxygen atom (red), all the others are attached to carbon and can not hydrogen bond. This can influence the local conformation of the polypeptide, indeed residues such as serine and asparagine are known to adopt conformations which most other amino acids cannot. Furthermore, this group can form a hydrogen bond with another polar group by donating or accepting a proton (a table showing donors and acceptors in polar and charged amino acid side chains can be found at the foldit site. For clarity, one serine is shown (in magenta) in b) [pdb:
Racemic serine can be prepared in the laboratory from methyl acrylate in several steps: Web thanks to their polarity, water molecules happily attract each other. One one of the three ammonium hydrogens can form a hydrogen bond with only one of the two oxygen atoms. Tyrosine possesses a hydroxyl group in the aromatic ring, making it a phenol derivative. Web thus, hydrogen bonds always contain three atoms and only those three. The opposite behavior is seen for cysm, as this residue seems to prefer more exposure than cys to the solvent. A survey of known protein structures reveals that approximately 70% of serine residues and at least 85% (potentially 100%) of threonine residues in helices make hydrogen bonds to carbonyl oxygen atoms in the preceding turn of the helix.
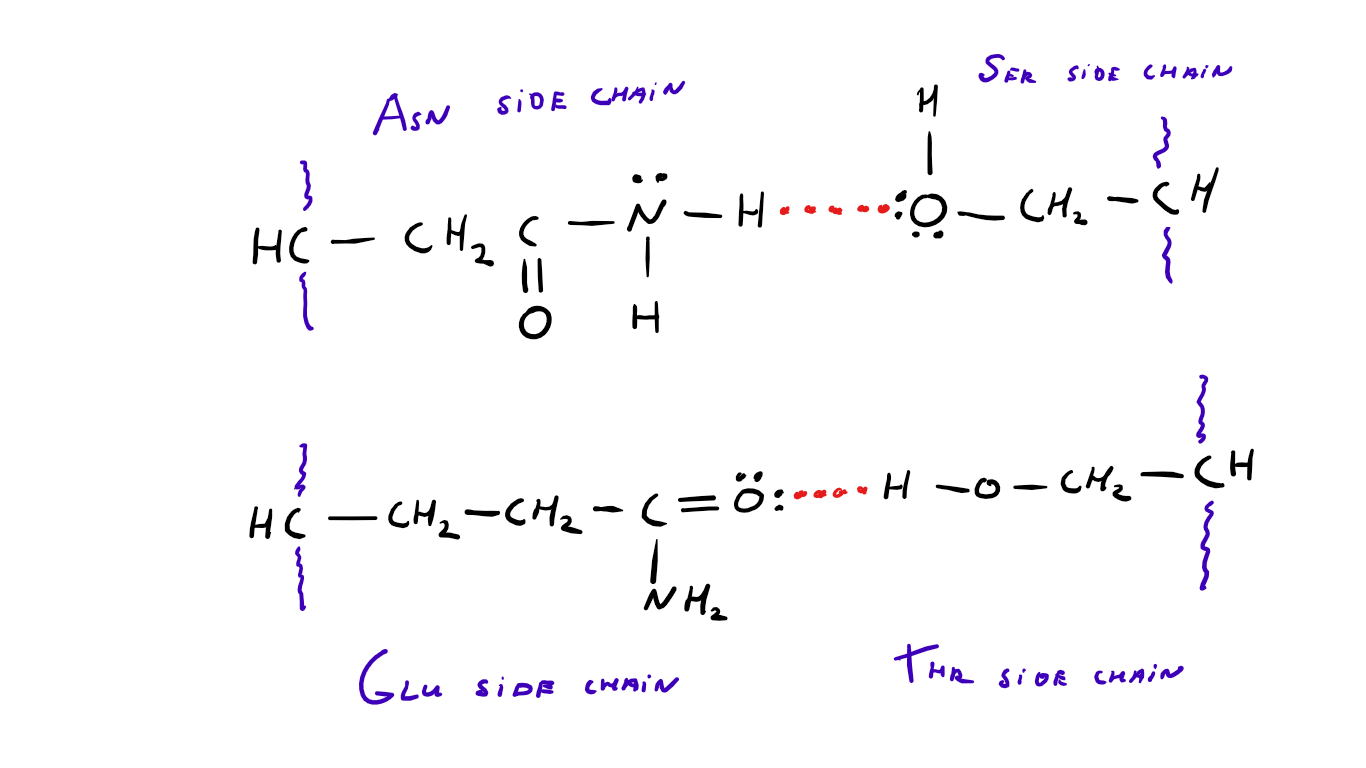
Web serine and threonine possess hydroxyl groups in their side chains and as these polar groups are close to the main chain they can form hydrogen bonds with it. Web the hydrogen on the oh group in serine can act as a hydrogen bond donor as it is slightly positive (delta positive) and the oxygen on the oh group in serine can act as a hydrogen bond acceptor as it is slightly negative (delta negative). Web lots of amino acids contain groups in the side chains which have a hydrogen atom attached to either an oxygen or a nitrogen atom. A survey of known protein structures reveals that approximately 70% of serine residues and at least 85% (potentially 100%) of threonine residues in helices make hydrogen bonds to carbonyl oxygen atoms in the preceding turn of the helix. The high frequency of intrahelical hydrogen bonding is of particular significance for intrinsic.
Web two amino acids, serine and threonine, contain aliphatic hydroxyl groups (that is, an oxygen atom bonded to a hydrogen atom, represented as ―oh). Answer only one, the one at the very top which is attached to the highly electrongative oxygen atom (red), all the others are attached to carbon and can not hydrogen bond. Web the hydrogen on the oh group in serine can act as a hydrogen bond donor as it is slightly positive (delta positive) and the oxygen on the oh group in serine can act as a hydrogen bond acceptor as it is slightly negative (delta negative). Web desolvation of the donor and the acceptor must occur for the hydrogen bond to form, such that the effects of hydration and hydrogen bond formation nearly cancel out. Web lots of amino acids contain groups in the side chains which have a hydrogen atom attached to either an oxygen or a nitrogen atom. Intermolecular forces (imfs) occur between molecules.
Hydrogenation of serine gives the diol serinol: Below is the structure of the amino acid, serine. Hoch 2 ch(nh 2)co 2 h + 2 h 2 → hoch 2 ch(nh 2)ch 2 oh + 2 h 2 o biological function metabolic Four hydrogen atoms in the compound can form hydrogen bonds. For example, the amino acid serine contains an.
The high frequency of intrahelical hydrogen bonding is of particular significance for intrinsic. Web the hydroxyl group is fairly reactive, being able to form hydrogen bonds with a variety of polar substrates. Four hydrogen atoms in the compound can form hydrogen bonds. Web examples of polar residues that form hydrogen bonds to an adjacent strand that extends further than its neighbour, including serines in b) the pancreatic ribonuclease family [pdb:
A Survey Of Known Protein Structures Reveals That Approximately 70% Of Serine Residues And At Least 85% (Potentially 100%) Of Threonine Residues In Helices Make Hydrogen Bonds To Carbonyl Oxygen Atoms In The Preceding Turn Of The Helix.
The opposite behavior is seen for cysm, as this residue seems to prefer more exposure than cys to the solvent. Web lots of amino acids contain groups in the side chains which have a hydrogen atom attached to either an oxygen or a nitrogen atom. This is a classic situation where hydrogen bonding can occur. • ser residues in membrane proteins are less solvent exposed while cys are more.
These Attractions Are An Example Of Hydrogen Bonds, Weak Interactions That Form Between A Hydrogen With A Partial Positive Charge And A More Electronegative Atom, Such As Oxygen.
Web thus, hydrogen bonds always contain three atoms and only those three. This can influence the local conformation of the polypeptide, indeed residues such as serine and asparagine are known to adopt conformations which most other amino acids cannot. Web ser is more solvent exposed and usually engages in strong hydrogen bonds. For example, the amino acid serine contains an.
One One Of The Three Ammonium Hydrogens Can Form A Hydrogen Bond With Only One Of The Two Oxygen Atoms.
Web thanks to their polarity, water molecules happily attract each other. The high frequency of intrahelical hydrogen bonding is of particular significance for intrinsic. Answer only one, the one at the very top which is attached to the highly electrongative oxygen atom (red), all the others are attached to carbon and can not hydrogen bond. Thus, a primary question in molecular design should be which donors and acceptors need to be satisfied and not how more hydrogen bonds can be formed.
The Plus End Of One—A Hydrogen Atom—Associates With The Minus End Of Another—An Oxygen Atom.
Hydrogenation of serine gives the diol serinol: Tyrosine possesses a hydroxyl group in the aromatic ring, making it a phenol derivative. Web examples of polar residues that form hydrogen bonds to an adjacent strand that extends further than its neighbour, including serines in b) the pancreatic ribonuclease family [pdb: Web the hydrogen on the oh group in serine can act as a hydrogen bond donor as it is slightly positive (delta positive) and the oxygen on the oh group in serine can act as a hydrogen bond acceptor as it is slightly negative (delta negative).