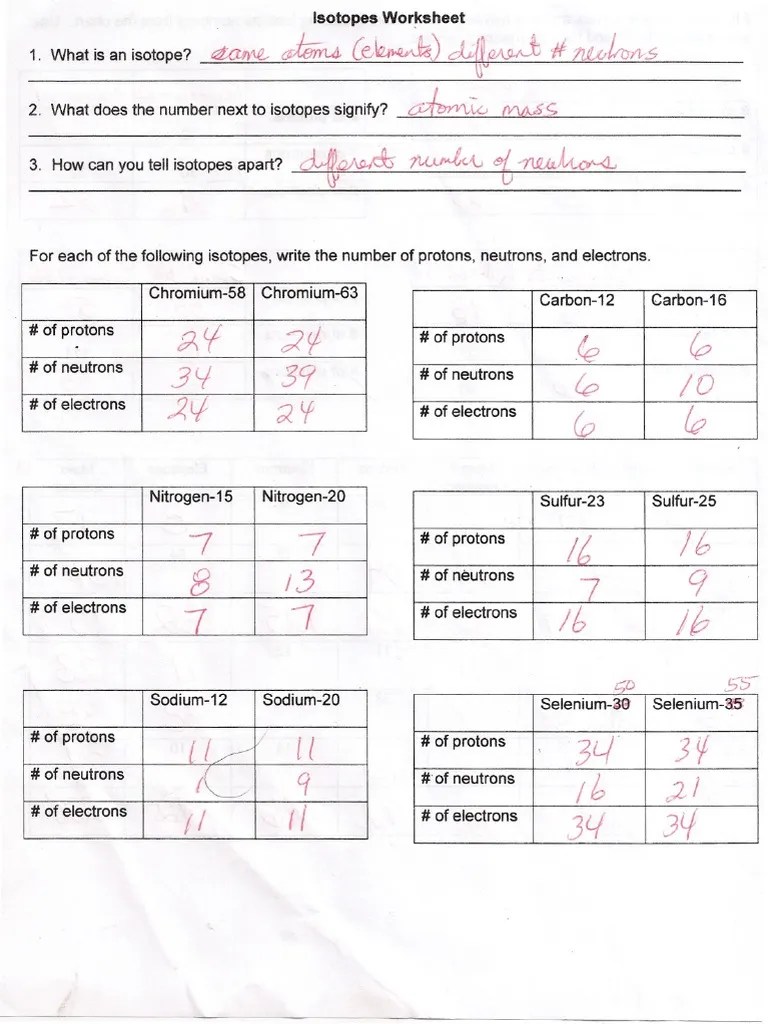
Atomic Structure And Isotopes Worksheet Answers
Atomic Structure And Isotopes Worksheet Answers - Each structure may be use d. An atom of potassium has an atomic number of 19 and a. Web docx, 430.5 kb. Calculate average atomic mass and isotopic abundance. 3.1.7 oxidation, reduction and redox equations. The mg 2+ ion has 12 neutrons in its nucleus; The atomic number of a magnesium atom is 12 and its mass number is 24. 81w 36 kr sld 37 kdu. (3) electron (1) neutron (1) proton (1) b) what is the atomic mass of this atom? The other worksheets are available for the atomic structure topic, including the topic as a whole, in my other resources.
The mg 2+ ion has 12 neutrons in its nucleus; If we know the number mass number and the atomic number, we can calculate the number of neutrons in the atom using: Describe the general arrangement of subatomic particles in the atom electrons surround the nucleus; Atoms of the same element with different numbers of neutrons are called isotopes. Click the card to flip 👆. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. Calculate average atomic mass and isotopic abundance.
(2) b neutrons (1) c protons (1) 2. Atoms of the same element with different numbers of neutrons are called isotopes. If we know the number mass number and the atomic number, we can calculate the number of neutrons in the atom using: Or isotopes, taking into account the _____ of each isotope. Includes lesson powerpoint, all tasks and all answers and technician request sheet (1st slide of powerpoint if applicable)
The atomic number of a carbon atom is 6 and its mass number is 12 The number of subatomic particles in an atom can be. Isotope symbo 131 il s3 1 ##### us |he. Define isotope, relative atomic mass (ram) calculate ram from isoptope data. Isotopes of an element have: Fully resources lesson on ks4 atomic structure and isotopes.
So different isotopes have different mass numbers but the same proton number. State the periodic law and explain the organization of elements in the periodic table. The mg 2+ ion has 12 neutrons in its nucleus; One isotope has a mass number of 10 and the other isotope has a mass number of 11. Protons electrons neutrons isotope name
Protons electrons neutrons isotope name One isotope has a mass number of 10 and the other isotope has a mass number of 11. (3) electron (1) neutron (1) proton (1) b) what is the atomic mass of this atom? Each structure may be use d.
Did This Video Help You?
The lesson pack covers atoms, elements and isotopes. All substances are made of tiny particles of matter called atoms which are the building blocks of all matter. 3.1.7 oxidation, reduction and redox equations. Define the unified atomic mass unit and average atomic mass.
Define Isotope, Relative Atomic Mass (Ram) Calculate Ram From Isoptope Data.
Never trust an atom, they make up everything! Number of neutrons = 12. Each atom is made of subatomic particles called protons, neutrons, and electrons. Understand the relationship between the masses of isotopes and the atomic weight of an element;
Atomic Structure P Ast Paper Q Uestions Science Exams Sorted 9.
Web basic atomic structure worksheet answers. Answer the following questions about these structures. The atomic structure and the periodic table resource is the first lesson in the gcse atomic structure and the periodic table unit. 81w 36 kr sld 37 kdu.
Chemistry Atomic Structure Practice I 17 Isotopic Symbol Na Potassium Name Symbol Z A #P #E #N † Z Ax Iron 53
Calculate the average atomic mass of chlorine if its isotopes and % abundances are as follows. The mg 2+ ion has 12 neutrons in its nucleus; Each structure may be use d. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells.